Accessioned in 2007.
=ATCC 12600 =ATCC 12600-U =CCM 885 =CCUG 1800 =CIP 65.8 =DSM 20231 =HAMBI 66 =IAM 12544 =NBRC 100910 =NCAIM B.01065 =NCCB 72047 =NCDO 949 =NCIMB 13062 =NCTC 8532.
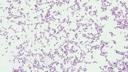
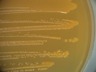
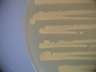
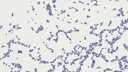
Staphylococcus aureus subsp. aureus.
Type strain [596,13170].
Medium: 12, 22; Temperature: 37°C; Rehydration fluid: 663. more information + Full / Short version of the cataloguepage top

open link in new window
open link in new window






 Instructions for an order
Instructions for an order